Measles, Mumps And Rubella Vaccine Live, Attenuated, (Freeze-Dried) M.M.R
Product description
Pharmaceutical Form: Lyophilised active component to be reconstituted with excipient diluent before use
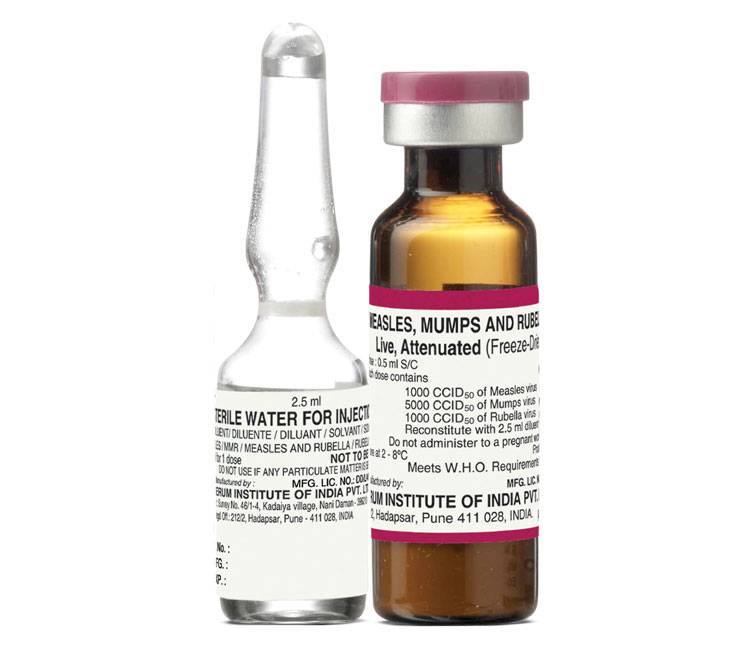
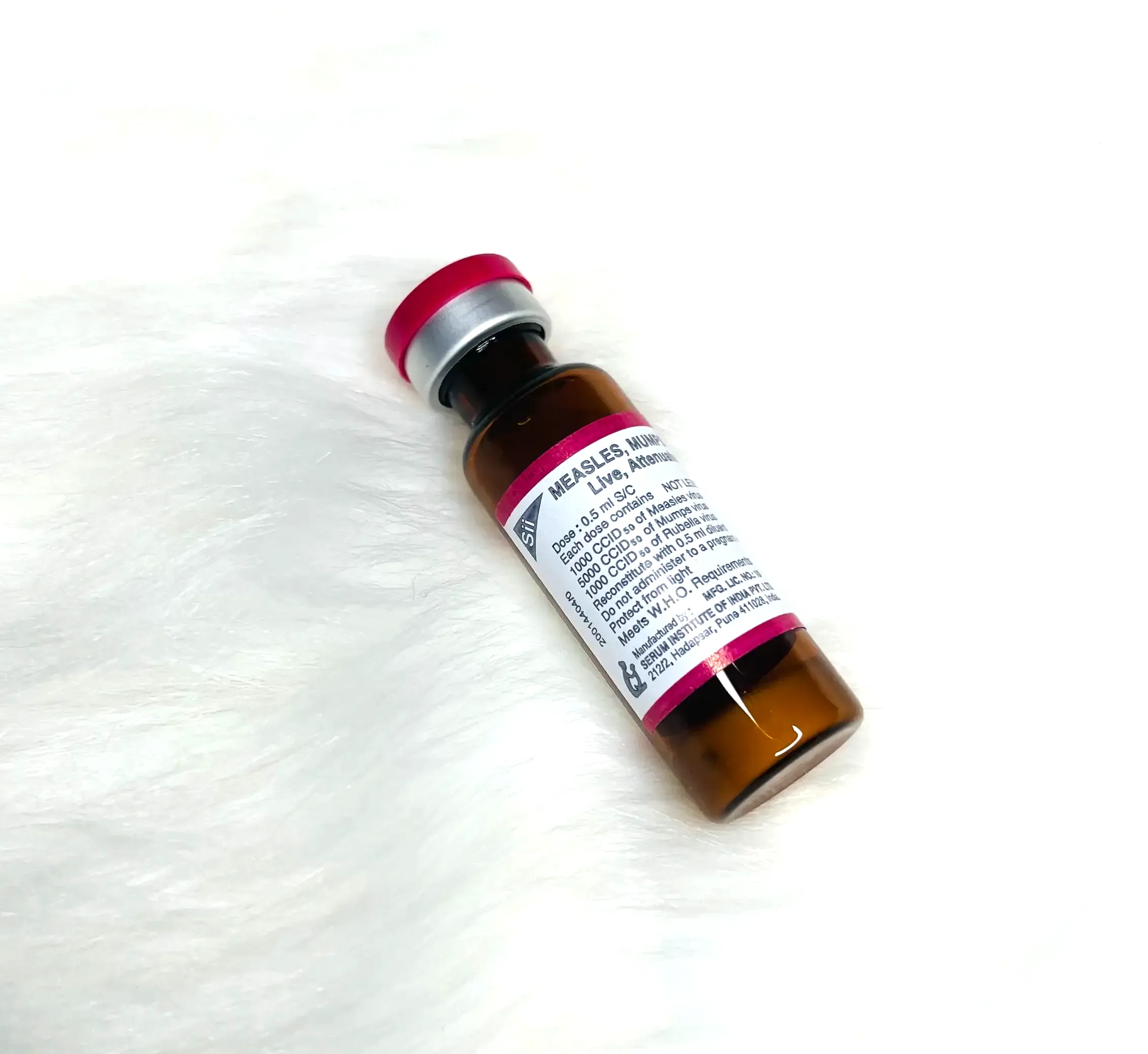
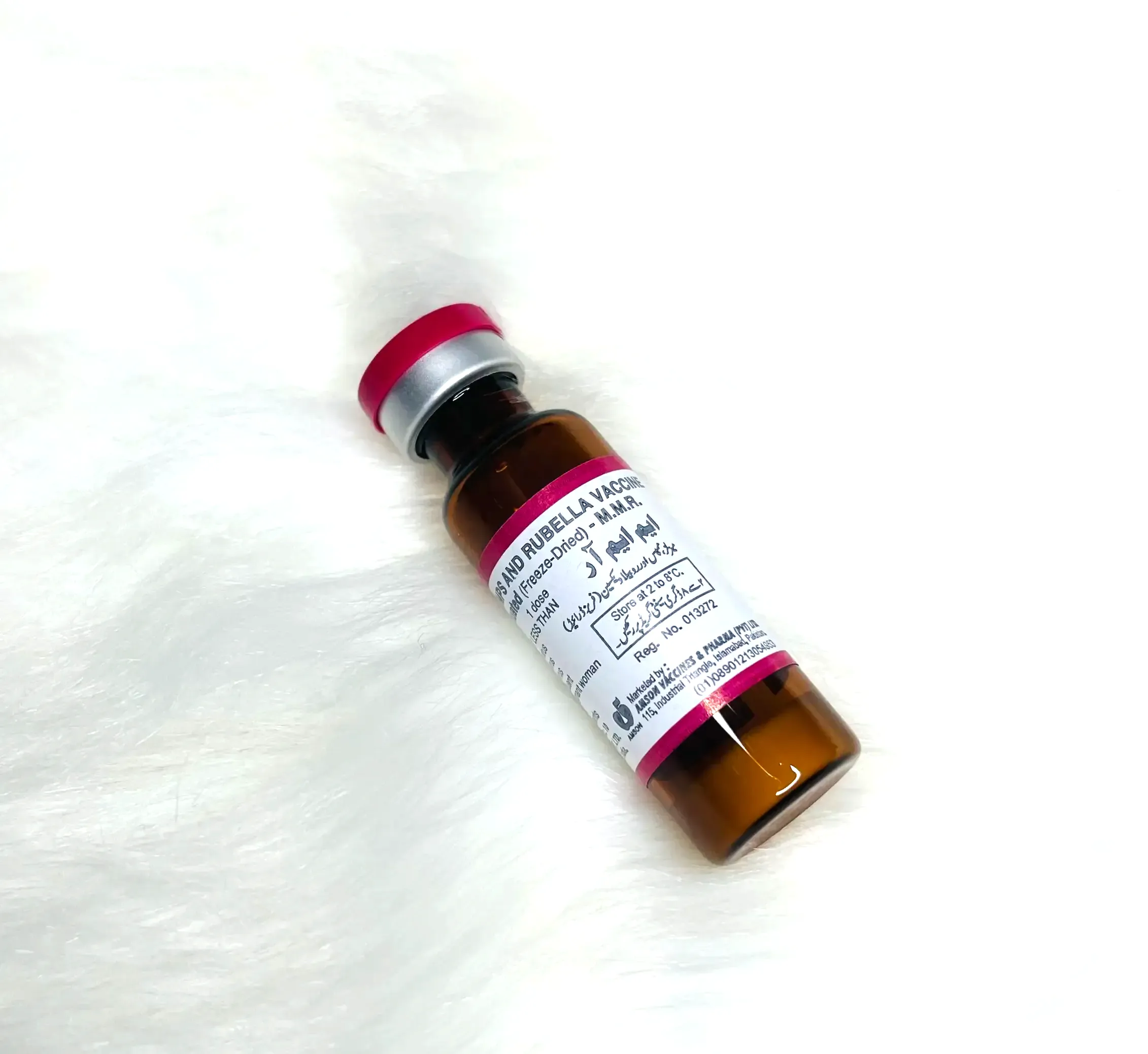
Presentation: Vial
Number of Doses: 5
Diluent: Water for Injection
Route of Administration: Subcutaneous
- Generic name: Measles, Mumps And Rubella Vaccine Live, Attenuated, (Freeze-Dried)
- Indication: For active immunization against measles, mumps and rubella in individuals 12 months of age, children and adolescents. Second dose of MMR is advocated any time before the age of 6 years (elementary school entry 4-6 years) and thereafter Measles and Rubella is recommended. Revaccination may seroconvert primary failures or boost antibody titres of previously vaccinated individuals whose titers have declined. The Advisory Committee on Immunization Practices (ACIP) recommends administration of the first dose of MMR at 12-15 months of age and administration of the second dose of MMR at 4-6 years of age. Immunization of susceptible non-pregnant adolescent and adult females is indicated if certain precautions are observed (see CONTRAINDICATIONS). The vaccine can be safely and effectively given simultaneously with DTP, DT, TT, Td, BCG, Polio vaccine (OPV and IPV), Haemophilus influenza type b, Hepatitis B, Yellow fever vaccine and vitamin a supplementation.



















Reviews
There are no reviews yet.